Distributed Pharmaceutical Analysis Lab
The Distributed Pharmaceutical Analysis Lab (DPAL) provides high quality, validated chemical analysis of pharmaceutical samples from partners in the developing world. This collaboration with Chemists Without Borders uses analytical resources at academic institutions in the US to perform characterization of suspect medications that have been collected and screened in Kenya, Uganda, Tanzania, India, Nepal, or Malawi.
How it works:
DPAL participants must demonstrate that their HPLC system is suitable for pharmaceutical assays by performing a suite of tests, such as linearity, accuracy, precision, and matrix spike-recovery. If all goes well, this will take 40-45 injections, and makes a good instrumental analysis or undergrad research lab project. Once the assay is approved, we will ship you a box of samples for analysis and work with you and your students to follow up with the regulatory agency if necessary.
1) . Register for Open Science Framework at osf.io (it is free and they don't spam you!) and contact Marya (mlieberm@nd.edu) or Max (mwilfing@nd.edu) to pick a pharmaceutical.
2) . We open a project folder for your institution at the Open Science Framework DPAL site. The project site hosts the most current methodology for analyzing different drugs and Excel spreadsheets for entering data. You can register your data at any time, which creates an unique DOI leading to a web archive showing the data at the point you write a report or prepare a publication/presentation.
3) File a system suitability report. DPAL provides standard HPLC methods and procedures and standard samples for validating the methods on your instruments. After you find your system's linear range for pure API, we will ship you expired samples to test matrix recovery.
4) . Once you have shown that your system and sample preparation method is suitable for the analysis of the target pharmaceutical, we will ship you samples collected in Kenya or other developing countries. You analyze the samples and perform the QC checks and return the analytical data and your conclusions to DPAL.
5) Suspicious samples undergo additional analysis at UND and then we jointly write a report to alert the appropriate country medical regulatory authorities and sometimes, the WHO.
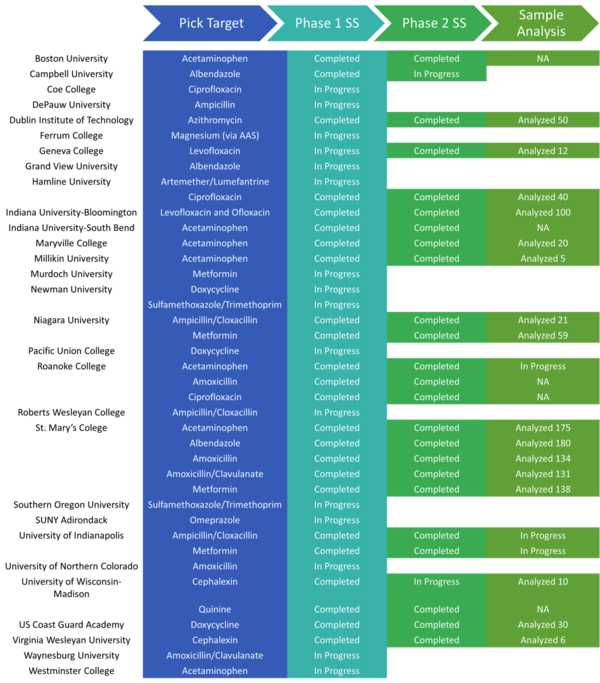
System validation and analysis of the samples can be incorporated into undergraduate research, a chemistry club activity, or into formal coursework such as an instrumental analysis or analytical chemistry lab. As of Jan 2020, 68 poster presentations, 38 oral presentations, and 28 senior thesis or capstones have been completed by DPAL participants at 29 colleges and universities.
Download 2023 HPLC Methodology Manual [PDF, 1MB]
This manual is a work in progress. Questions and comments are welcome. The first sections describe how to choose and validate HPLC assays; suggested assay conditions for several antibiotics are included at the end of the manual. Other assays may be found in pharmacopeia monographs, such as the WHO's International Pharmacopoeia and the USP monographs. DPAL participants may contact Dr. Lieberman for more information about USP monograph procedures (they are behind a paywall, but USP has kindly given us access through their Academic Connection Program).
Data Processing Templates for Excel and a wiki on HPLC troubleshooting are available on the Open Science Framework project site for DPAL members.
How to get started:
A faculty member must be formally responsible for managing the project and taking custody of the samples. Contact Prof. Marya Lieberman to discuss this process. You can register for OSF at osf.io.
A good starting point is to download the methodology manual (use the link above) and check the availability of a suitable HPLC system and columns; most C18 columns are suitable for amoxicillin or ampicillin analysis.
Depending on your location, technical assistance with repairing or testing HPLCs may be available through Chemists without Borders.
Why we're doing this
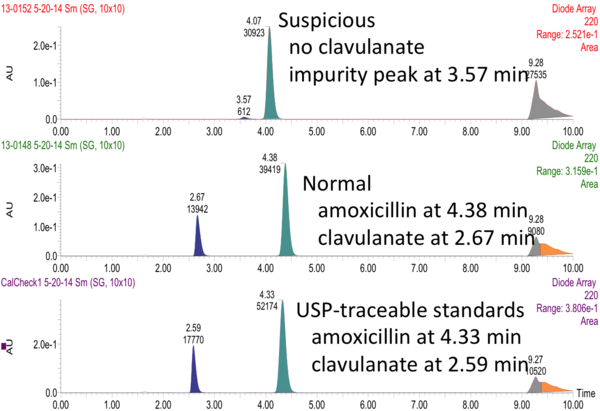
The suspicious sample of amoxicillin/clavulanate below was discovered during an HPLC training session for summer undergraduate researcher Rebecca Ryan. Clavulanate is a potentiating agent that enables amoxicillin to kill resistant bacteria. Thanks to the efforts of undergraduate researchers at DPAL, this sample was reported to the Kenyan medical regulatory authorities and the World Health Organization RapidAlert system in 2014; the product was quarantined in Kenya. The origin of the degradation appears to have been a packaging problem. Subsequent batches of this manufacturer's product corrected the packaging flaw.
Samples
We have over 1,200 samples of antibiotics, such as amoxicillin, from eastern Africa and SE Asia available for analysis. A new project has brought samples of chemotherapy adjacent medications such as MESNA, hydroxyurea, and leucovorin that will be available in 2024-2027.
