Happy new year from the PAD team at Notre Dame!
 Back row: Sarah B, Meg B, Madeline S, Margaret B, Meghanne T, Nils O. Front row: Galen B, Rachel M, Tracy L-C, Dr. L, Jessica Z
Back row: Sarah B, Meg B, Madeline S, Margaret B, Meghanne T, Nils O. Front row: Galen B, Rachel M, Tracy L-C, Dr. L, Jessica Z
 Back row: Sarah B, Meg B, Madeline S, Margaret B, Meghanne T, Nils O. Front row: Galen B, Rachel M, Tracy L-C, Dr. L, Jessica Z
Back row: Sarah B, Meg B, Madeline S, Margaret B, Meghanne T, Nils O. Front row: Galen B, Rachel M, Tracy L-C, Dr. L, Jessica Z
The Medicine Quality and Public Health conference at Oxford University had 200 participants from over 50 countries, including the Directors General of the Tanzanian and Nigerian drug regulatory agencies and academics, NGOs, WHO, and pharmaceutical companies.
Marya Lieberman and Philip Coyne attended…
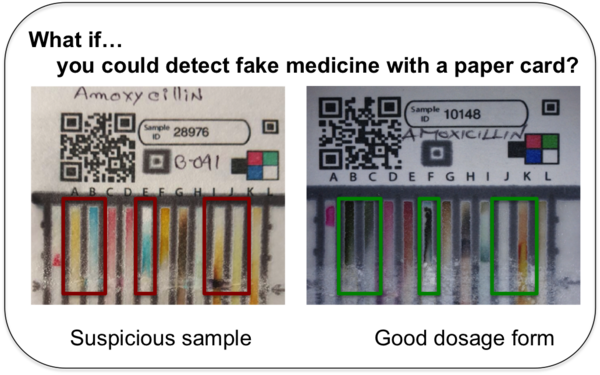
At the Oxford MQPH conference, Marya Lieberman presented in a session titled Screening Technologies: Will they Save the Day? The talk was captured by the sketch artist. Here we see a summary of how the PAD works and the results of the supply chain exercise that the 20 students in the short course…
https://news.nd.edu/news/scientists-take-aim-at-illicit-supply-chain-networks-of-fake-medications/
The National Science Foundation has awarded our team an exploratory grant to use PADs to detect and dismantle supply chains for bad quality pharmaceuticals. We will work with Professor Karen Smilowitz at…
Check out the NPR interview with Muhammad Zaman about his book "Bitter Pills: The Global War on Counterfeit Drugs". He is the BU prof who developed a portable dissolution testing device
Our proposal titled "The whole world is watching: tracking bad quality medicine with citizen science and an electronic ledger" was funded by the Notre Dame Blockchain Working Group for 2018! Computer scientists James and Chris Sweet will work with the PAD project to institute a distributed electronic…
Notre Dame has licensed the PAD technology to Veripad, LLC. This startup company has developed a cell-phone based PAD reader that automatically analyzes the PAD image for the user. Watch for more developments!
…
by Zoie Alexiou
The World Health Organisation (WHO) has recently published an advocacy document in attempt to quantify the cost…