Pharmaceutical Alerts
Warning: Three lots of a drug stated to be Cisteen cisplatin for injection, obtained from Tikkur Anbesa Specialized Hospital in Addis Ababa, Ethiopia. This product was substadard .

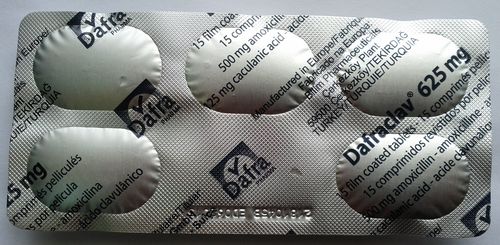
Warning: This package is stated to be Dafraclav 625 mg, containing amoxycillin and clavulanic acid, and was purchased in the Eldoret region in 2015. On physical examination of the package, the name of one of the active ingredients was found to be mis-spelled on the packet, which is a violation of section 3.2.2.7.1.1 of the Kenyan guidelines for the registration of pharmaceutical products. The KPPB has been notified and we are attempting to contact the manufacturer. Posted May 5, 2016
Update: May 15, 2016; the manufacturer's representative replied that the lot number (2181045B) is an officially released batch number and that the printing error is known to their quality control department. He pointed out that the drug was expired at time of purchase. We'll follow up on this with the PPB. We commend the manufacturer for a fast and factual response.
Alert: Bactoclav-625 lot numbers NMBBV0002, NMBBV0008, NMBBV0015, NMBBV0020, NMBBY0005, BTBBV0036, and BTBBV0024, claimed to be produced by Micro Labs; samples were collected in Kisumu region in 2013-2014.
This product is a potentiated beta lactam antibiotic stated to contain both amoxycillin and clavulanate. Analysis of single tablets by HPLC showed little or no clavulanate in multiple tablets taken from several packages of the products. Absence of this active pharmaceutical ingredient could render the pills medically ineffective.
A full report has been filed with the KPPB and with the WHO Rapid Alert system. KPPB is seeking additional samples from these lot numbers for further analysis.
Posted 22 June, 2015
Alert: Bactoclav-625 lot BTBBV0024, claimed to be produced by Micro Labs; Bactoclav-625 lot BTBBV0036, claimed to be produced by Micro Labs; samples were collected in Western Kenya in 2012-2013.
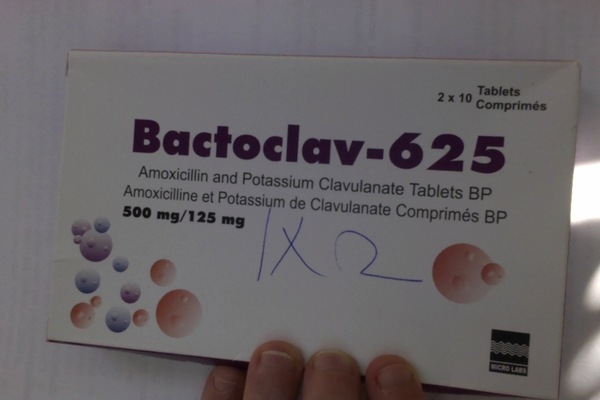
This product is a potentiated beta lactam antibiotic stated to contain both amoxycillin and clavulanate. Analysis of single tablets by HPLC showed little or no clavulanate in multiple samples taken from several packages of the products. Absence of this active pharmaceutical ingredient could render the pills medically ineffective.
A full report has been filed with the KPPB and with the WHO Rapid Alert system.

Posted 6 October, 2014